 
Radioactivity was examined further by Marie Curie and Pierre Curie, for which they earned the Nobel Prize in Physics in 1903. Quantum tunneling was developed from the study of radioactivity, which was discovered in 1896 by Henri Becquerel. Its acceptance as a general physical phenomenon came mid-century. The effect was predicted in the early 20th century. Tunneling limits the minimum size of devices used in microelectronics because electrons tunnel readily through insulating layers and transistors that are thinner than about 1 nm. Tunneling applications include the tunnel diode, quantum computing, flash memory, and the scanning tunneling microscope. Tunneling plays an essential role in physical phenomena such as nuclear fusion and alpha radioactive decay of atomic nuclei. 
Some sources describe the mere penetration of a wave function into the barrier, without transmission on the other side, as a tunneling effect. Tunneling is readily detectable with barriers of thickness about 1–3 nm or smaller for electrons, and about 0.1 nm or smaller for heavier particles such as protons or hydrogen atoms. The probability of transmission of a wave packet through a barrier decreases exponentially with the barrier height, the barrier width, and the tunneling particle's mass, so tunneling is seen most prominently in low-mass particles such as electrons or protons tunneling through microscopically narrow barriers. Tunneling is a consequence of the wave nature of matter, where the quantum wave function describes the state of a particle or other physical system, and wave equations such as the Schrödinger equation describe their behavior. In the published 1927 paper, Heisenberg originally concluded that the uncertainty principle was Δ pΔ q ≈ h using the full Planck constant.In physics, quantum tunnelling, barrier penetration, or simply tunnelling is a quantum mechanical phenomenon in which an object such as an electron or atom passes through a potential energy barrier that, according to classical mechanics, the object does not have sufficient energy to enter or surmount. Introduced first in 1927 by German physicist Werner Heisenberg, the uncertainty principle states that the more precisely the position of some particle is determined, the less precisely its momentum can be predicted from initial conditions, and vice versa. The uncertainty principle implies that it is, in general, not possible to predict the value of both paired variables’ quantities with arbitrary certainty beyond certain limit, in which a trade-off (frequency-position trade-off) between both appears, even if all initial conditions are specified and known. Such paired-variables are, therefore, known as complementary variables or canonically conjugate variables and, depending on interpretation, the uncertainty-principle limits to what extent such conjugate properties maintain their approximate meaning, as the mathematical framework of quantum physics does not support the notion of simultaneously well-defined conjugate properties expressed by a single value. 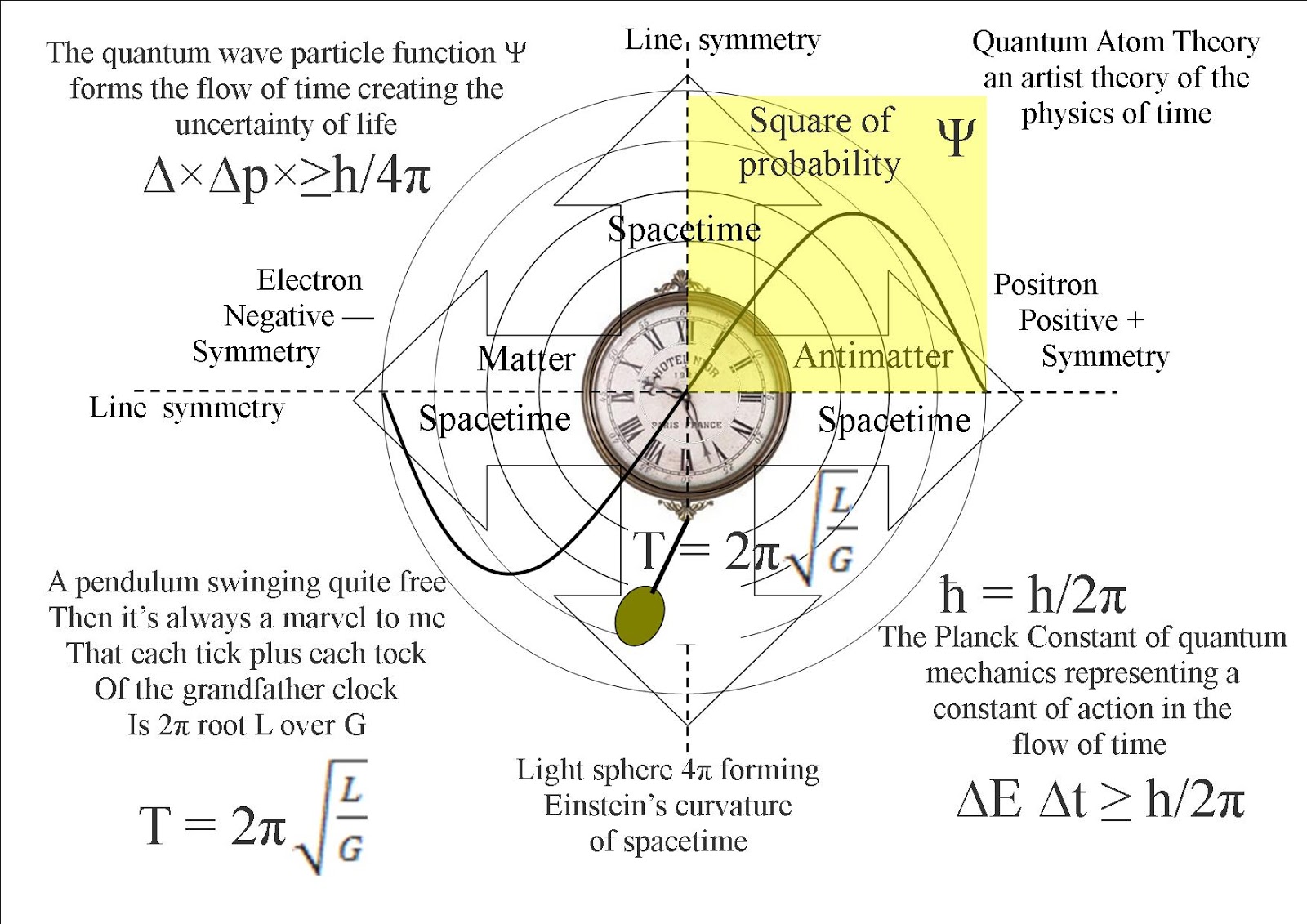
In quantum mechanics, the uncertainty principle (also known as Heisenberg's uncertainty principle) is any of a variety of mathematical inequalities asserting a fundamental limit to the accuracy the values for certain related pairs of physical quantities of a particle, such as position, x, and momentum, p, can be predicted from initial conditions appearing a trade-off between them. Uncertainty principle of Heisenberg, 1927. Canonical commutation rule for position q and momentum p variables of a particle, 1927. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
